|
Because CO 2 is a linear molecule, our dipoles are symmetrical their magnitudes are equal but their orientations are opposite.ĭispersion forces are caused by the attraction between nearby molecules. Nonpolar molecules are those that have an even charge distribution but no dipole moment. An asymmetrical molecule, on the other hand, lacks a symmetry axis and can be rotated. In chemistry, molecular symmetry refers to the symmetry found in molecules as well as the classification of these molecules based on their symmetry.Ī symmetrical molecule is one whose appearance does not change when rotated around a symmetry axis the original and rotated states are indistinguishable. The following are significant words used by the London Forces: Symmetrical 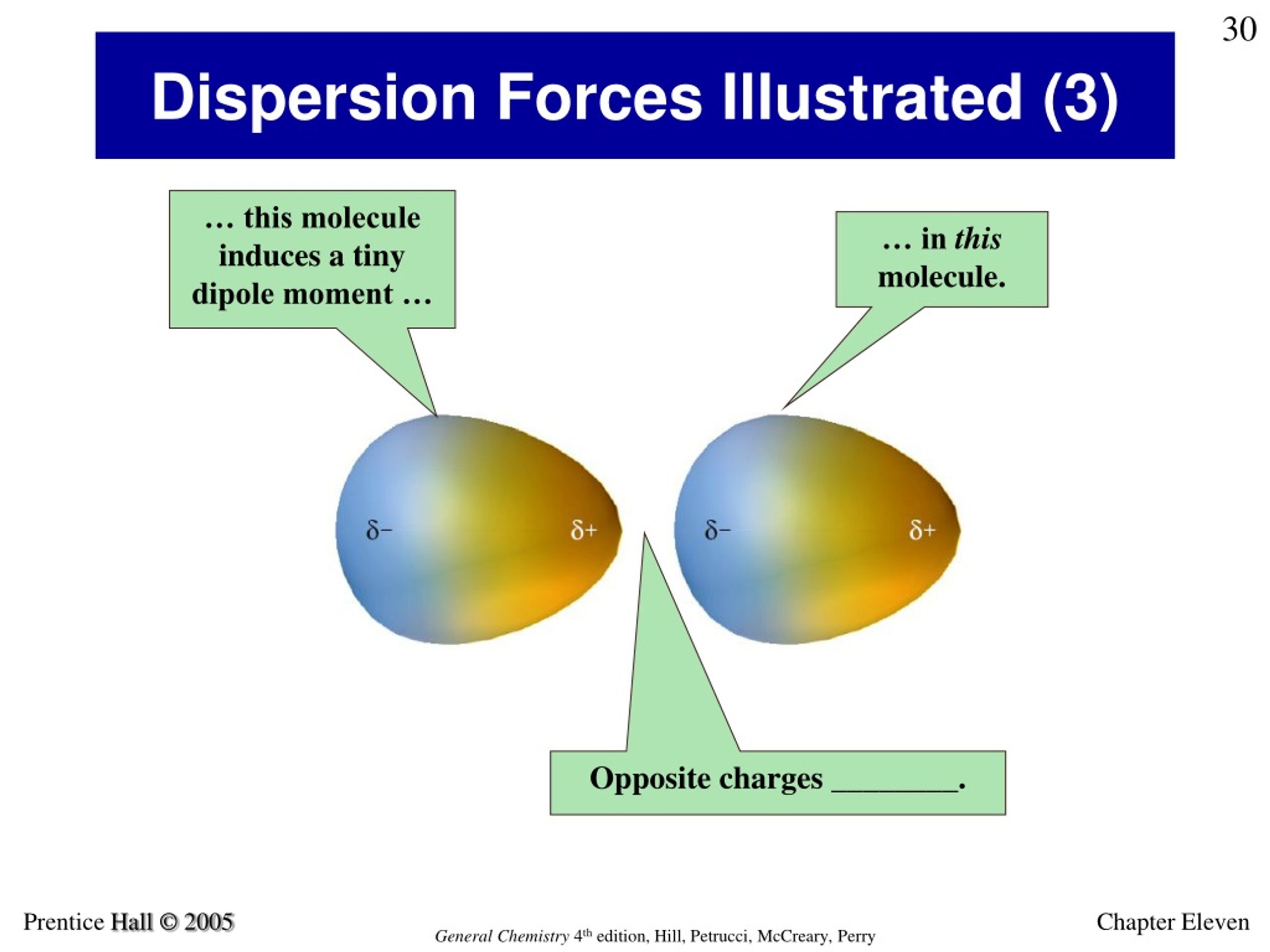
We begin with a review of many famous inorganic and organometallic complexes whose dispersion forces have been explicitly detected or calculated, and then move on to recent examples of organic species that have influenced contemporary thinking. The physical and chemical properties of organometallic and inorganic compounds are influenced by these attractive forces, which are mainly between C–H moieties in auxiliary ligands. The role of light atoms, such as hydrogen, has been generally overlooked in discussions of molecule stability and reactivity. 
Small, easily polarised molecules, such as water molecules, are an exception. Despite its weakness, the dispersion force is frequently the most prominent of the three van der Waals forces. The London dispersion force is the weakest of the van der Waals forces, and it’s what causes nonpolar atoms or molecules to condense into liquids or solids as the temperature drops.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
